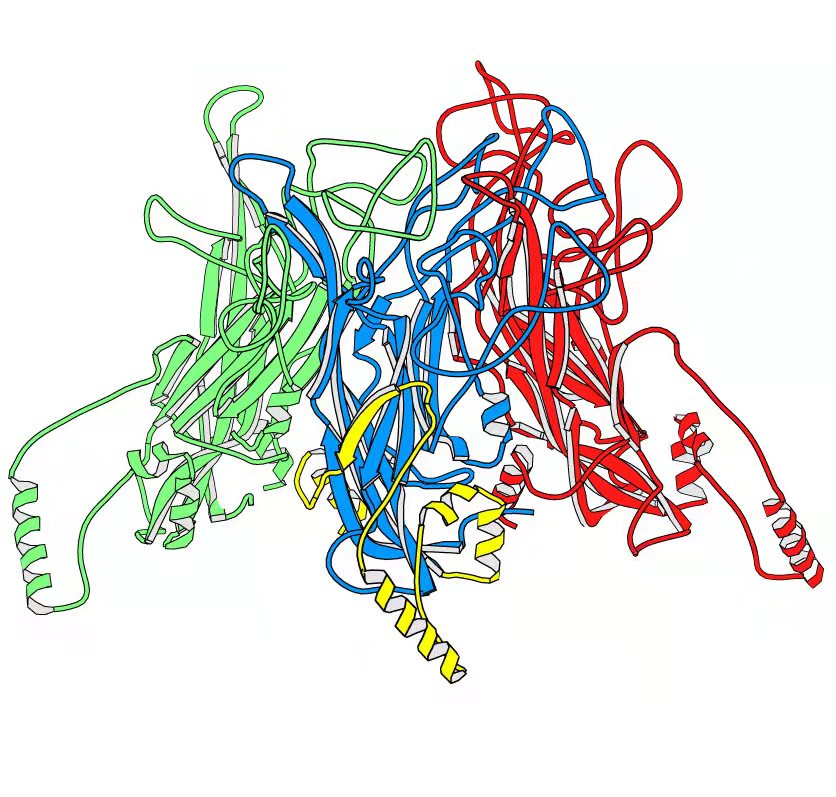
基于结构的抗原设计平台
蛋白的结构决定了其功能。“基于结构的抗原设计”是利用蛋白结构信息来设计疫苗候选物,使其拥有最佳的理化性质、生物学活性和药效等,其优势在于可以利用原子水平的蛋白结构信息来精准改造和优化抗原。我们已建立成熟的基于结构的抗原设计技术平台,可从蛋白的一级序列、二级结构和三维结构等水平进行分析,设计出具有理想性质的目标抗原。我们基于蛋白结构设计了HPV疫苗系列产品、重组二价新冠疫苗和呼吸道合胞病毒疫苗的抗原。
蛋白的结构决定了其功能。“基于结构的抗原设计”是利用蛋白结构信息来设计疫苗候选物,使其拥有最佳的理化性质、生物学活性和药效等,其优势在于可以利用原子水平的蛋白结构信息来精准改造和优化抗原。我们已建立成熟的基于结构的抗原设计技术平台,可从蛋白的一级序列、二级结构和三维结构等水平进行分析,设计出具有理想性质的目标抗原。我们基于蛋白结构设计了HPV疫苗系列产品、重组二价新冠疫苗和呼吸道合胞病毒疫苗的抗原。
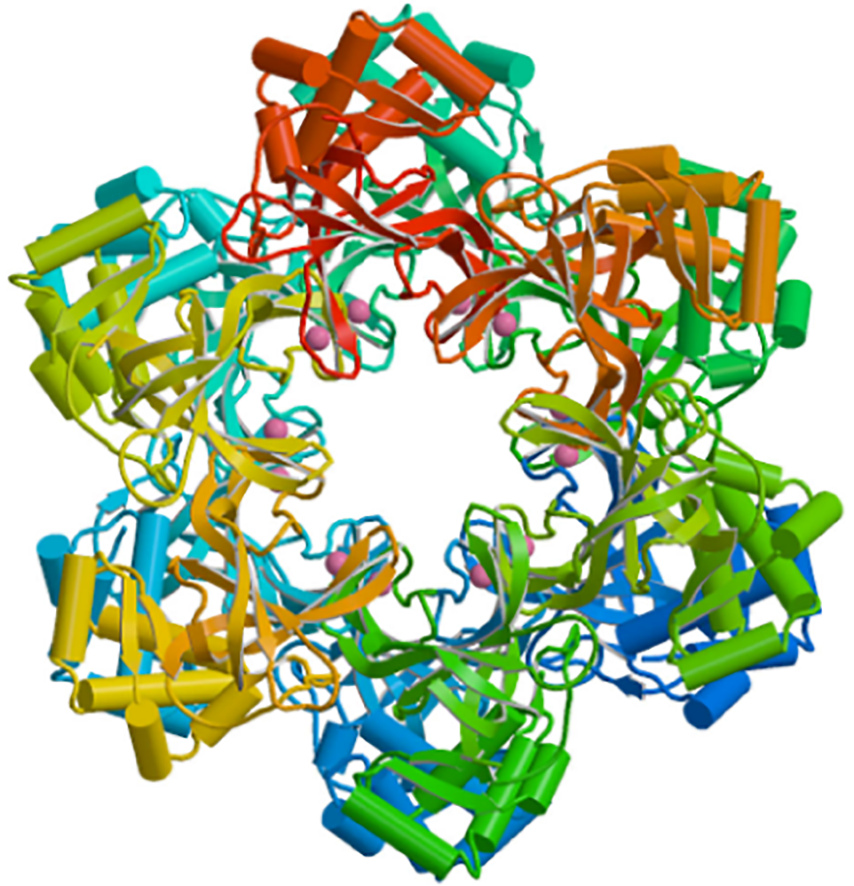
由于蛋白种类繁多、性质各异、结构复杂,蛋白表达有时非常棘手。成功的产品开发依赖于使用正确的蛋白表达系统,同时需根据表达系统对目标蛋白编码序列进行相应优化及对表达载体进行适当改造。我们已建立成熟的基因工程和蛋白表达技术平台,涵盖大肠杆菌、酵母和CHO细胞表达体系,适用各类重组疫苗产品开发。基于本技术平台,我们已成功改造和高水平表达了HPV疫苗系列产品、重组二价新冠疫苗和重组多价诺如病毒疫苗的抗原。

重组疫苗制备工艺复杂,各个工艺步骤都高度专业化,需要配备相应的专业人才和设备设施。同时,疫苗属高度监管的行业,生产过程需要符合国家法律法规及行业监管政策要求,因此需要投入大量资金建设符合监管要求的生产场所。公司已成功搭建疫苗工程化技术平台,建设了符合中国和欧盟GMP条件的3,000+平方米疫苗生产中试车间,配备了完备的生产、质控及制剂设备设施。公司开发阶段的产品均可在该中试车间完成工艺放大,有助于未来顺利向商业化生产基地进行技术转移。
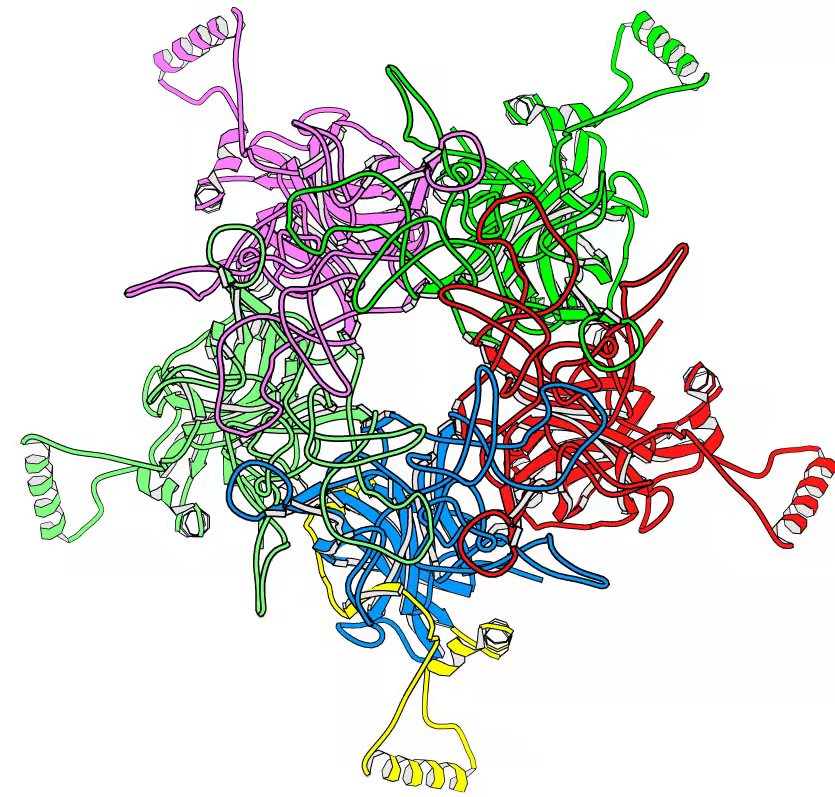
疫苗产品的质量控制环节中对疫苗效力的评价极为重要。疫苗效力评价方法的选择应和所评价疫苗的作用机制相关,且可在临床前测试中挑选出免疫原性好的候选疫苗。疫苗效力评价方法包括体内效力试验和体外相对效力试验等,其建立过程充满挑战,原因包括疫苗独特的成分、多价性质和长生命周期等。我们已搭建成熟的重组疫苗效力评价技术平台,包括假病毒制备和检测方法、结合阻断抗体检测方法及双抗体夹心酶联免疫法,并已成功应用于HPV疫苗、重组呼吸道合胞病毒疫苗和重组多价诺如病毒疫苗的开发中。